Since December 14, Muskoka Paramedic Services has been running a pilot to help determine the efficacy of a rapid COVID-19 test.
“[We] will go for 8 weeks as part of the Phase 1 pilot. Phases 2 & 3 will involve other participants and not MPS,” explained chief of Muskoka Paramedic Services (MPS), Jeff McWilliam. “The purpose is to gather data that will support the overall efficacy/accuracy of this rapid test so it can be determined if it should be part of the province’s ongoing testing strategy.”
The pilot is being conducted on behalf of the Ministry of Health, Ontario Health, and Public Health Ontario. Participants in the pilot can range from frontline workers and businesses to healthcare workers, those in congregate settings, and paramedic services, according to McWilliam.
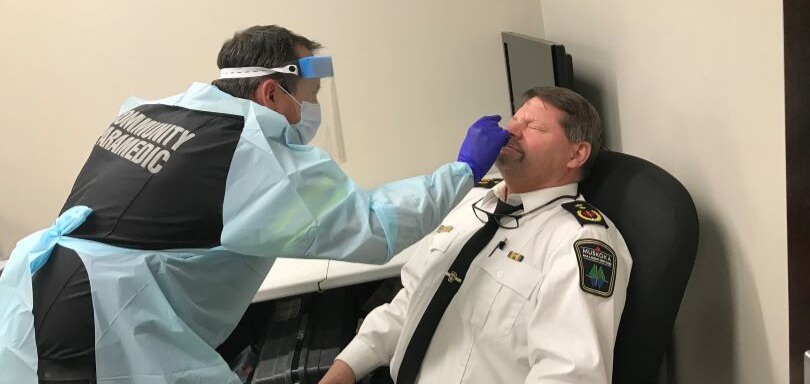
Here in Muskoka, the Panbio antigen testing pilot project is being conducted on asymptomatic Muskoka Paramedic Services staff who volunteer.
The Panbio rapid antigen detection test—which was developed by Abbott Rapid Diagnostics—is conducted through a nasopharyngeal swab. Antigen testing detects specific proteins from the virus “to screen and identify people who need further testing,” according to Ontario’s Ministry of Health. While rapid testing can deliver results in less than 15 minutes, the tests are less sensitive than laboratory-based tests and may produce false negatives, particularly in asymptomatic people. The Panbio test appears to be highly specific, however, according to reports from both the company and early studies, making it unlikely to produce a false positive.
The Panbio COVID-19 antigen rapid test was authorized by Health Canada in October, the first antigen rapid test the federal agency had approved.
Health Canada has noted that rapid testing could be beneficial for outbreak control or monitoring in populations where there is high community prevalence of the virus, monitoring high-risk congregate living or work settings, or in remote communities where standard testing is not available.
According to McWilliam, 38 tests have been conducted so far and to date none of them have returned a false negative or positive for COVID-19.
“As this is a new test pilot, data specifically related to Panbio’s accuracy as a screening tool is primarily from lab-based studies, which may not be entirely reflective of ‘real world’ experience. We are proud to be part of this trial and believe that this tool could be powerful moving forward, however sensitivity and specificity ranges need to continue to test high during the pilot project in order to implement broadly,” noted McWilliam via email.
Don’t miss out on Doppler!
Sign up here to receive our email digest with links to our most recent stories.
Local news in your inbox three times per week!
Click here to support local news

0 Comments